top of page

Relating Lattice, Solution & Hydration Enthalpies
Physical
A2
Enthalpy
AQA Content
You should know:
Hess's Law states that the overall enthalpy change for a reaction is independent of the route taken.
Energy cannot be created or destroyed but can be transferred.
Exothermic processes release energy (heat) - energy is available to do work.
Endothermic reactions require energy with heat taken into a system.
Specification Notes
Breaking a bond always requires energy so it’s always an endothermic process and the enthalpy change is positive (∆H is +).
Forming bonds is an exothermic process and the enthalpy change is negative (∆H is –).
Notes
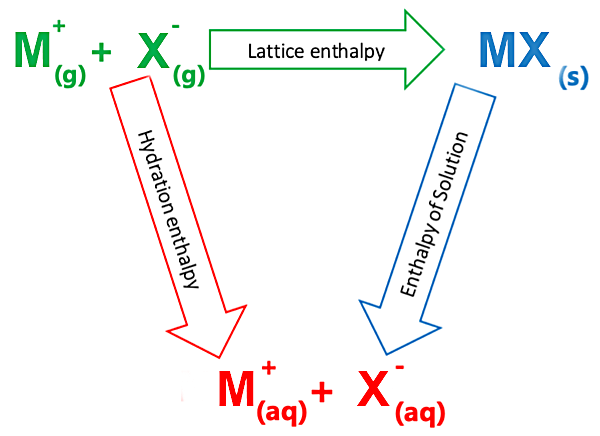
Here we look at the relationship between the lattice enthalpy, enthalpy of solution and hydration enthalpy
Some Enthalpy Definitions
Here's a link to other definitions of enthalpy change...
Taking sodium chloride as an example, let's look at dissolving in a little more detail...
When the salt dissolves in water the ions are separated - separating any particle is an endothermic process. Also, water molecules need to separate from each other to be able to dissolve the sodium chloride. Remember, water molecules have hydrogen bonds, some of which need to be overcome to solvate the sodium chloride ions. The enthalpy change is positive since it’s an endothermic process.
When sodium chloride is dissolved, water molecules surround the sodium cation and chlorine anion through ion-dipole interactions. Each of these bonds release energy - an exothermic process since ion-dipole bonds are formed. Gaseous sodium and chloride ions associate with water and turn into the aqueous state - solvated by water.
The enthalpy change of solution is equal to the negative enthalpy change of the lattice energy plus the enthalpy change of hydration. Hydration involves the separated ions solvated by water and some of the hydrogen bonds of water being broken to get separated water molecules. Remember - lattice energy is always negative (exothermic) – dissociation is therefore endothermic.
Remember
1. Lattice Enthalpy is always negative
2. Enthalpy of hydration is always negative
3. The magnitude and sign of the enthalpy of solution depends on the value of 1 and 2 above
Born-Haber Cycle for sodium chloride
RED PATH = BLUE PATH
To quickly calculate the enthalpy of solution:
Add the negative enthalpy change of the lattice energy to the enthalpy of hydration.
Walkthrough Examples:
Example 1:
Calculate the enthalpy change of solution for calcium fluoride given the following data
Example 2:
The enthalpy of hydration for NaCl is –783 kJ/mol. The lattice energy is –787 kJ/mol. Calculate the enthalpy of solution for NaCl(s).
You could also take the negative value of the lattice enthalpy and add it to the hydration enthalpy:
Note:
When solid sodium chloride is completely dissolved in water, the temperature of the solution should decrease slightly since the salt takes away some of the thermal energy from the surrounding water.
Example 3:
Calculate the enthalpy of solution of Mg(OH)2 given that:
Advice for students:
Develop a clear stepwise method for tackling these problems
Separate different mathematical operations by using brackets
Look carefully at the moles of ions involved - you may need to multiply some of the values (see Walkthrough Examples 1 and 3 above)
Links
bottom of page